Can Girls Have Duchenne?
PPMD is often asked the question: can girls or women have Duchenne? This is a complex question to answer. In rare cases, girls can have symptoms that follow the same pattern and progression that we see in classic Duchenne muscular dystrophy. This degree of severity could occur for several uncommon genetic reasons, such as the girl having highly skewed X-inactivation (only the X chromosome with the DMD gene variant is turned on throughout the body), having only one copy of the DMD gene, or having a DMD gene variant on both copies of the X chromosome. Whether a female child should be given a diagnosis of Duchenne or Becker depends on a combination of factors which include symptoms, test results, and careful interpretation by a specialist experienced with muscular dystrophy.
PPMD believes that young female carriers in our community deserve the same opportunities to receive optimal care and inclusion in research studies as boys with Duchenne or Becker. We have chosen to use the term females with dystrophinopathy to include girls in the “spectrum” of dystrophinopathies that include Duchenne and Becker. By emphasizing the spectrum of dystrophinopathies, PPMD can raise awareness of the health concerns for everyone affected and better inform healthcare professionals, researchers, and family members. We are optimistic these discussions among experts in the field will continue to drive improvements in the diagnosis, care, and awareness of females with dystrophinopathy.
WHAT SYMPTOMS CAN GIRLS HAVE?
Females with dystrophinopathy can have a wide variety of symptoms that also vary in severity. Symptoms may include:
- Muscle weakness
- Fatigue or low endurance
- Impaired balance and frequent falls
- Muscle pain or cramps
- Cognitive and behavioral diagnoses (ADHD, learning disabilities, etc.)
- Cardiac changes (fibrosis or scarring seen on cardiac MRI, changes in function seen on echo)
Why do some young girls have symptoms of Duchenne or Becker but others do not?
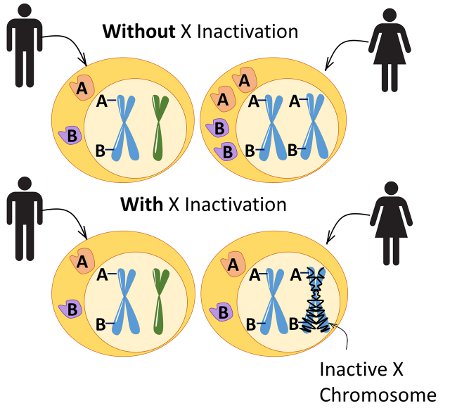
The dystrophin (DMD) gene is located on the X-chromosome. Because boys typically only have only one X-chromosome (and one Y-chromosome), they typically only have one copy of the DMD gene. Thus, if this copy of the gene isn’t making enough dystrophin protein, the boy will have Duchenne or Becker muscular dystrophy. However, girls typically have TWO X-chromosomes, which means they typically have TWO copies of the DMD gene.
In every cell of the female body, usually one copy of the X chromosome is “turned off”, which is a process called X-inactivation. For most girls and women, this process is random and their body will have roughly a 50:50 mixture of cells with one copy of the X chromosome turned off versus the other. However, less commonly, X-inactivation can be “skewed”, and one copy is turned off a majority of the time compared to the other. For a girl or woman who is a carrier of a DMD gene variant on one X chromosome, her degree of symptoms can be influenced by skewed X-inactivation. For example, a girl who is a carrier but has the clinical symptoms of Duchenne might have skewed X-inactivation in which, for reasons that aren’t usually known, her body is consistently turning off the normal copy of the gene. In this situation, the only copy of the DMD gene available to the body is the one that isn’t making the dystrophin protein, and that is why the symptoms of Duchenne appear.
In the past, neuromuscular specialists ordered X-inactivation testing to try and understand why symptoms are happening in female carriers. However, we have learned that X-inactivation testing is not accurate because X-inactivation can vary among muscle groups and other tissues, causing asymmetrical symptoms such as one leg being weaker than the other. Specialists suggest that clinical symptoms, rather than X-linked inactivation testing, are currently the most appropriate way to assess whether a female is affected with dystrophinopathy.
MY DAUGHTER DOES NOT HAVE SYMPTOMS. SHOULD SHE STILL HAVE GENETIC TESTING?
The decision of when to test a young asymptomatic female is a family decision and should focus on the maturity of the person being tested as well as the needs of the parents. A discussion with your pediatrician, genetic counselor, or other healthcare provider would also help to weigh the pros and cons of testing an asymptomatic individual.
Early detection of the DMD gene variant may lead to earlier and better treatment in symptomatic girls. PPMD advocates for better education about the potential health risks of being a female with dystrophinopathy. If symptoms are identified, then genetic testing is recommended.
CARE FOR YOUNG FEMALES WITH DYSTROPHINOPATHY
Similar to males, females should be treated based on symptom severity. Young girls with any symptoms described above should be evaluated by a comprehensive neuromuscular clinic, preferably a Certified Duchenne Care Center, for evaluation and to determine an appropriate care plan. If the child’s symptoms match the pattern of Duchenne muscular dystrophy, their care may be planned to follow the standard care guidelines for Duchenne.
CAN GIRLS PARTICIPATE IN CLINICAL TRIALs?
In the past, females were not included in most research studies. However, this is evolving with time and in recent years, some research studies have included females with dystrophinopathy. For more information, visit our clinical trials page. Females with dystrophinopathy can also participate in The Duchenne Registry, which is an online, patient-report research program managed by PPMD.






