Variant-Specific Therapies
It is an incredibly exciting time in the Duchenne and Becker muscular dystrophy community with so many potential therapies in the pipeline. Many of these therapies are variant-specific, meaning they will only work for individuals with certain changes (often called variants or mutations) in their Duchenne gene.
This brief explanation may help you to understand if you or your child may be a candidate for one of the variant-specific therapies that is approved or in development.
If you haven’t had genetic testing, or need repeat testing to confirm if you are eligible for a variant-specific therapy, PPMD’s Decode Duchenne program provides free genetic testing to eligible individuals. If you have questions, please contact the Decode Duchenne team.
Two key types of variant specific therapies
There are currently two types of variant-specific therapies in clinical trials for Duchenne: nonsense variant suppression and exon skipping.
Nonsense variant suppression
Individuals with Duchenne who have a nonsense variant may be candidates for nonsense variant read-through. A nonsense variant is a single change in the Duchenne gene that creates a “premature stop codon”, which basically stops production of the dystrophin protein too early in the process and results in a non-functioning protein. Some (but not all) genetic test reports will state if the variant is a nonsense variant. Learn more about nonsense variant read-through.
Exon skipping
Individuals with Duchenne who have certain “out-of-frame” deletions may be candidates for exon skipping. Out-of-frame deletions usually result in little or no dystrophin protein which usually means the individual will have Duchenne. With exon skipping, one or more of the exons bordering the deletion are also skipped, with the purpose of turning the “out-of-frame” deletion into an “in-frame” deletion. In-frame deletions usually result in some dystrophin protein production. However, the impact that exon skipping will have on an individual’s disease progression has varied depending on the type of exon skipping therapy used. Newer “next generation” exon skipping therapies are also being developed. Learn more about exon skipping.
Determining if a deletion is amenable to exon-skipping therapies
If you know your/your child’s genetic variant is an exon deletion, our Exon Deletion Tool can help you understand if you/your child may be a candidate for an exon skipping therapy.
There are more than 150 different deletions of one or more exons in the dystrophin gene that can cause Duchenne muscular dystrophy. Exon-skipping research and development is at different stages for many of these deletions. Exon-skipping therapy for some deletions has been FDA-approved or is in clinical trial. Therapies are approved for skipping of exons 51, 53, and 45, and therapies for skipping of exon 44 are in clinical trial. Researchers are also focusing on skipping of other exons in the hot-spot region, including exons 50 and 52. The pie chart below shows the percentage of the Duchenne population who are amenable to exon skipping and which exons would benefit the highest percentage of the population.
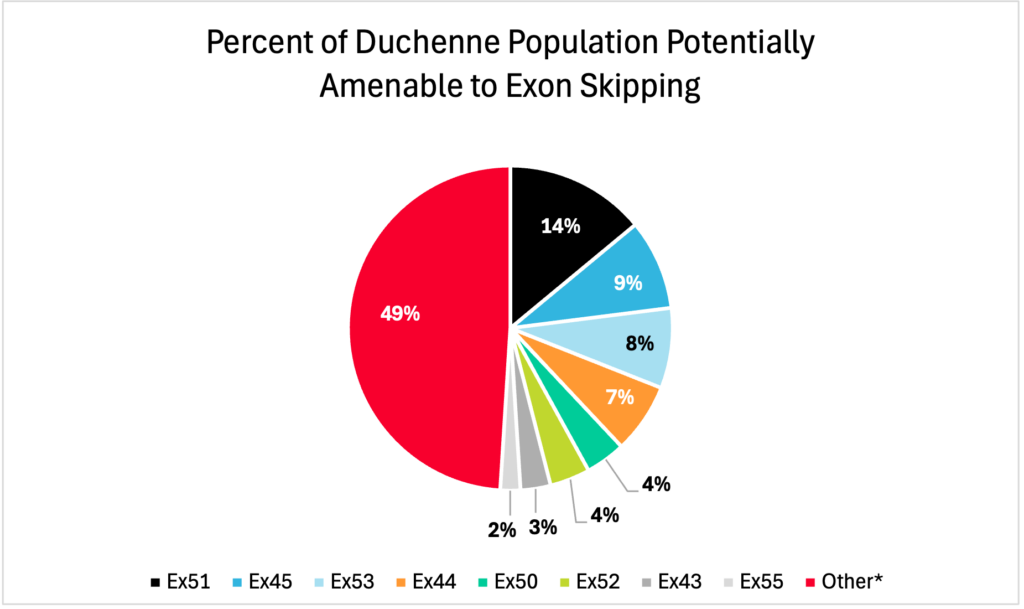
*Other = May be amenable to skipping other exons or may not be amenable to any exon skipping
Note that not all deletions will be amenable to exon skipping. This includes deletions that would need more than one exon skipped or that are in specific regions of the gene that may not be correctable with exon skipping. If you have questions about genetic results or variant-specific therapies, talk with your care team. You may also reach out to PPMD’s Registry & Genetics Team or schedule a PPMD for You appointment.
Looking to the future: other types of technologies
Researchers are studying other technologies that may provide more variant-specific therapies in the future. One of these technologies is called CRISPR/Cas9. The goal of this technique is to edit the gene itself to correct the variant causing disease. CRISPR/cas9 is being explored for many conditions, including Duchenne. To learn more about gene editing and gene therapy, visit PPMD’s Gene Therapy Hub.
Parent Project Muscular Dystrophy offers many ways to stay up to date on research advancements and clinical trials:






